Circulating atypical cells detected in blood of lung cancer patients
Warsaw, October 2021 – New study data presented at the European Congress of Cytology – held from 3-6 Oct in Warsaw, Poland – confirmed that X-ZELL is able to detect tumour-associated circulating atypical cells in the blood of lung cancer patients
The dataset comprised interim results from a pilot study carried out in Thailand by X-ZELL and Dr Kittipong Maneechotesuwan from Mahidol University’s Siriraj Hospital in Bangkok, and was presented by X-ZELL Founder & CEO, Dr Sebastian Bhakdi.
According to Dr Bhakdi, tumours start shedding atypical cells into the blood when they are only 1-2 mm in size. Being able to detect – and understand – these cells could be key to non-invasive early cancer detection.
In previous studies focusing on prostate cancer patients, X-ZELL was able to identify individual, tumour-associated atypical cells in 10mL blood and visualise them for AI-assisted analysis.
“It’s still early days but we are confident that our single-cell diagnostics technology is able to detect atypical cells in the blood that are linked to a lung carcinomas,” he explained. “After demonstrating the same in the blood of prostate cancer patients, this could be an important step towards establishing single-cell diagnostics as a powerful new tool in the fight against cancer.”
To access the ECC 2021 abstract, titled The immunocytomorphological landscape of tumour-associated circulating atypical cells in the blood of lung cancer patients, click here to download the official abstract book or scroll down to the bottom of the page for a preview.
To view the presentation, please contact X-ZELL via email.
Contact:
Sebastian Grote
Head of Strategy, Marketing & Communications
sebastian.g@x-zell.com
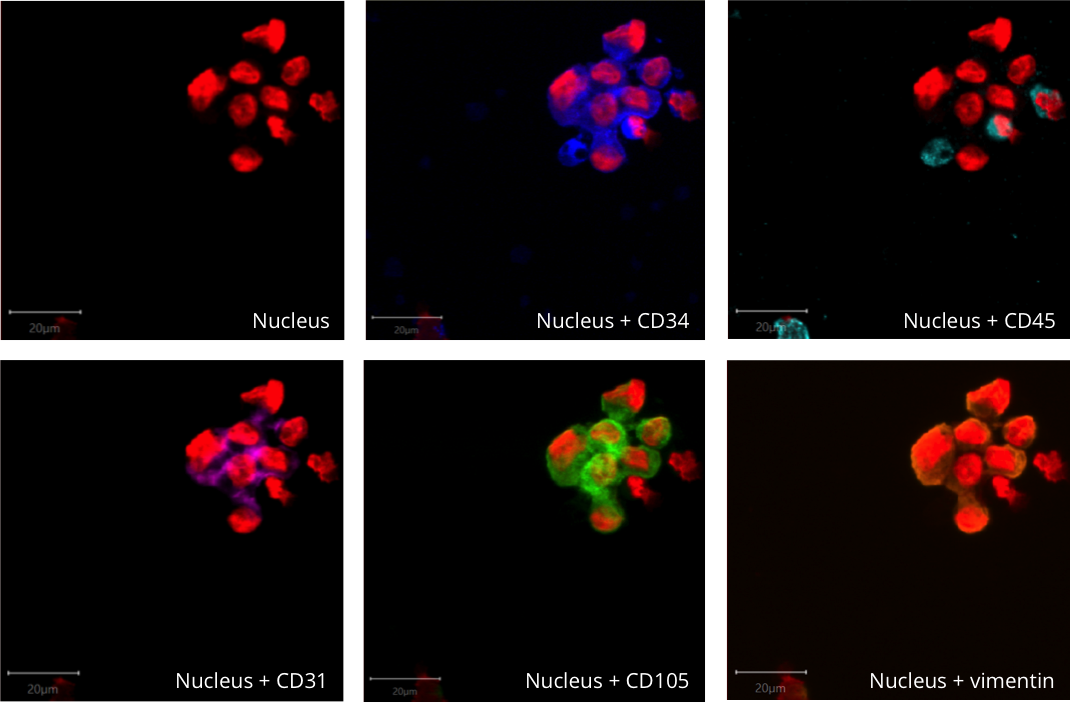
Image: Circulating atypical cell clusters. (c) X-ZELL 2021
Abstract: The immunocytomorphological landscape of tumour-associated circulating atypical cells in the blood of lung cancer patients
Department of Pathobiology, Faculty of Science, Mahidol University, Bangkok, Thailand and X-ZELL Biotech Pte. Ltd., Singapore
Krittika Teerapuncharoen, Department of Internal Medicine, Faculty of Medicine (Siriraj Hospital), Mahidol University, Bangkok, Thailand Kamontip Kunwipakorn, Department of Internal Medicine, Faculty of Medicine (Siriraj Hospital), Mahidol University, Bangkok, Thailand Chatchai Narula, International Diagnostics Co. Ltd., Bangkok, Thailand Ponpan Thaicharoen, International Diagnostics Co. Ltd., Bangkok, Thailand Kittipong Maneechotesuwan, Department of Internal Medicine, Faculty of Medicine (Siriraj Hospital), Mahidol University, Bangkok, Thailand.
Background
Populations of tumour-associated circulating atypical cells (tCAC) in the blood of solid-tumour patients still lack systematic characterization. Here we present an analysis of untouched rare-cell populations obtained from liquid biopsies of 47 patients with suspected lung cancer during the discovery phase of our ongoing study. (1–5)
Cubital vein blood was drawn before bronchoalveolar lavage (BAL) to avoid cross-contamination with cells potentially released into the circulation. Rare cells were isolated and characterized by red blood cell lysis, hMX high-gradient magnetic white blood cell (WBC) depletion, Cryoimmunostaining and eight-channel fluorescence microscopy. Liquid biopsy results were matched with BAL and lung biopsy pathology results.
Results
We defined tCAC as CD45-negative clumps with anisokaryosis or single cells with increased or decreased chromatin, multinucleation, and/or blast-like appearance with high nuclear-to-cytoplasmic ratio. Immunophenotypes of such tCAC comprised endothelial (vimentin+, CD31+, CD34+), epithelial (cytokeratin+), epithelial-mesenchymal transition (cytokeratin+, vimentin+) and unknown-CD45-negative. We also observed “atypical” WBC (hyperploidic, CD34+, CD45weak),“activated” WBC (nuclear enlargement, loose chromatin, CD45+), hematopoietic stem cells (CD34+, CD45weak) as well as varying numbers and phenotypes of circulating megakaryocytes. Following the above definition, 13 out of 24 patients with a positive lung tissue biopsy had tCAC. An additional two biopsy-positive patients showed atypical WBC. However, none of the 23 patients with negative biopsy showed any tCAC or atypical WBC, while activated WBC and megakaryocytes were regular observations in most patients. Interestingly, nuclear enlargement (loose chromatin), indentation, irregularity, binucleation or any particular immunophenotype alone were no clear predictors of malignancy in both CD45-negative and CD45-positive cells.
Conclusion
A fascinating and diverse landscape of tCAC circulates in the blood of lung cancer patients. Improved immunocytomorphological characterization of these rare-cell subpopulations may result in a novel cytological tool amenable for artificial-intelligence-based image analysis and eventually, for routine analysis of tCAC and improved diagnostics of lung cancer.
References
1. Ilie, M. et al. Current challenges for detection of circulating tumor cells and cell-free circulating nucleic acids , and their characterization in non-small cell lung carcinoma patients . What is the best blood substrate for personalized medicine ? 2, 1– 10 (2014). 2. Ilie, M. et al. Clinical value of circulating endothelial cells and of soluble CD146 levels in patients undergoing surgery for non-small cell lung cancer. Br. J. Cancer 110, 1236–43 (2014). 3. Mohan, S., Chemi, F. & Brady, G. Challenges and unanswered questions for the next decade of circulating tumour cell research in lung cancer. Transl. Lung Cancer Res. 6, 454– 472 (2017). 4. Guibert, N., Pradines, A., Mazieres, J. & Favre, G. Current and future applications of liquid biopsy in nonsmall cell lung cancer from early to advanced stages. Eur. Respir. Rev. 29, (2020). 5. Hofman, V., Heeke, S., Marquette, C. H., Ilié, M. & Hofman, P. Circulating tumor cell detection in lung cancer: But to what end? Cancers (Basel). 11, (2019).
© 2024 X-ZELL Biotech Pte. Ltd.
Find Us